Abstract
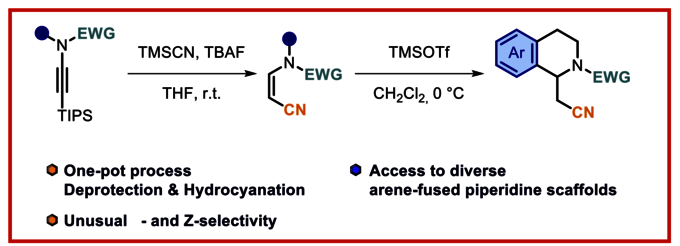
Metal-free β- and Z-selective hydrocyanation of terminal ynamides using TMSCN and TBAF is described. The transformation proceeds with excellent regio- and stereoselectivity, delivering exclusively Z-configured tertiary enamides. Treatment with TMSOTf promotes clean Z to E isomerization. Simultaneous activation of the nitrile moiety by TMSOTf in the presence of an internal nucleophile induces intramolecular cyclization, yielding piperidine frameworks. The reaction accommodates a range of nucleophiles, enabling the synthesis of structurally diverse and previously inaccessible heterocyclic scaffolds.


![[Translate to English:]](/websites/_processed_/0/4/csm_signature-unistra_fee3442f1d.png)
![[Translate to English:]](/websites/_processed_/0/e/csm_logo-cnrs_c0f610620b.png)
![[Translate to English:]](/websites/_processed_/9/4/csm_logo-fondation-lehn_24043a5484.png)