Abstract
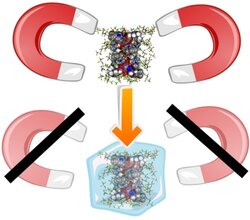
Gd5(L)16(H2O)8](Tf2N)15 was obtained from reaction of Gd2O3 with 1-carboxymethyl-3-ethylimidazolium chloride (LHCl). The material was found to be an ionic liquid that freezes to glassy state on cooling to −30 °C. Variable-temperature magnetic studies reveal the presence of weak magnetic intramolecular interactions in the glass. Isothermal variable-field magnetization demonstrates a magnetocaloric effect (MCE), which is the first finding of such an effect in a molecular glass. This MCE is explainable by an uncoupled representation, with a magnetic entropy change of −11.36 J K−1 kg−1 at 1.8 K for a 0–7 T magnetic field change, and with a refrigerant capacity of 125.9 J kg−1, in the 1.8-50 K interval.
Graphical Abstract
A GdIII-based ionic liquid exhibits a magnetocaloric effect in a frozen glass state.



![[Translate to English:]](/websites/_processed_/0/4/csm_signature-unistra_fee3442f1d.png)
![[Translate to English:]](/websites/_processed_/0/e/csm_logo-cnrs_c0f610620b.png)
![[Translate to English:]](/websites/_processed_/9/4/csm_logo-fondation-lehn_24043a5484.png)